| Service Level | Read Depth | Sample Input | Input Quantity* | Buffer |
|---|---|---|---|---|
| Standard | 2-5K reads/sample | Purified AAV capsids | 2-5 × 10¹¹ vector genomes | 200 µL of any common AAV formulation buffer or PBS 200 µL stabilized in 1X Zymo DNA/RNA Shield |
| Big | 15-20K reads/sample | |||
| Full flow cell | Up to 20M reads/flow cell |
*Note, you can submit a minimum of 1 × 10¹¹ vector genomes, but we recommend 2-5 × 10¹¹ for best results.
Purify your viral capsids.
We accept only purified viral capsids. Affinity resin or ultracentrifugation purified AAV samples perform best. At this time total cell lysate with or without detergent does not generate a useful number of reads in our AAV sequencing service.
Requirements for best results
- All samples must be free of cellular debris and must be treated with DNase or Benzonase before being shipped to us. We do not treat AAV samples with DNase prior to DNA extraction.
- Crude PEG-precipitated virus that has been resuspended in PBS or similar buffer is compatible with our system but requires clarification before sending material. Centrifuging at 3000 RCF for 10 minutes then recovering the supernatant into a new tube while avoiding the pellet is sufficient clarification.
- Our protocol is compatible with common AAV formulation buffers as well as simple PBS or TBS. Low concentrations of Mg²⁺, glycerol, and PF-68 do not interfere with extraction or sequencing. If the sample is in a high salt solution (e.g. neutralized elution buffer from an affinity purification) we recommend buffer exchanging into PBS or a similar common formulation buffer.
- For samples stabilized in 1X Zymo DNA/RNA Shield:
Submit 200 µL final volume of a minimum of 1x1011 vg of AAV in 1X final concentration Zymo DNA/RNA shield in PCR strip tubes. We recommend submitting 2-5x1011 vg since ITR-directed PCR quantitation tends to over-estimate intended AAV transgene titer.
Zymo Research sells at 2X concentrate (Product #R1200-25) that can be added at equal volume to 100 µL of any common virus storage buffer.
- For international shipments we are not able to accept samples from BSL2 organisms or above.
Prepare your samples.
Submit a minimum of 1.0 × 10¹¹ total vector genome (vg) copies of purified intact AAV viral capsids as determined by qPCR or ddPCR. For best results, we recommend sending 2-5 × 10¹¹ vg/sample. The most common cause for sequencing failure is insufficient DNA input.
Capsids can be sent in a volume of up to 200 µL of buffered saline or common AAV storage solutions. Please do not send larger volumes of sample since only 200 µl will be processed in our workflow.
For international AAV shipments there is an additional requirement to preserve and inactivate your cells, as live viral capsids cannot be imported into the US. The samples need to be specifically preserved in Zymo DNA/RNA Shield (2X Concentrate) before shipping.
Please note:
- We have noticed that commonly used ITR-directed qPCR primers tend to over-estimate vector genome content.
- If you have dilute AAV that cannot achieve 1.0 × 10¹¹ vg in 200 ul (i.e. <5 × 10¹¹ vg/mL), we can still process the samples but the returned read-depth will be lower than our target range. Please contact support@plasmidsaurus.com first before sending us low titer samples.
Label your tubes.
Make sure you are using the right 200 μL PCR strip tubes for your order.
- We have found that PCR strip tubes with individual swing-caps work best and are the least likely to leak. We recommend Eppendorf 0.2mL 8-Strip PCR Tubes with Hinged Flat Caps.
- Please do not send precious AAV samples in strip cap tubes like these. We have found that the lids leak even after being wrapped in parafilm!
- If your AAV sample is being sent from a CDMO or third party holding facility which cannot repackage the material into 200 μL PCR strip tubes, please notify support@plasmidsaurus.com when you send the samples. We can accommodate other packaging formats, but this may add to processing time.
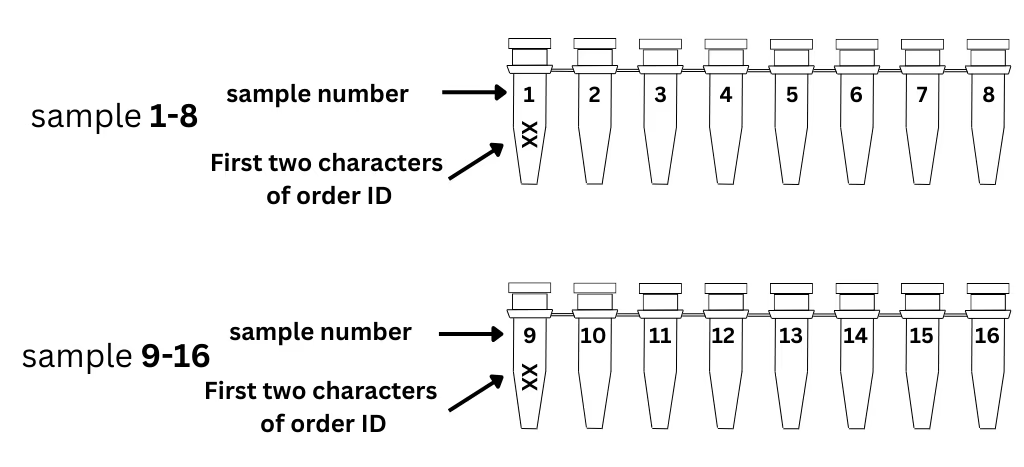
Set up your order online and receive your unique 6-character order ID.
Write the first 2 characters of your order ID on the 1st tube of each strip.
Package and ship your samples.
AAV samples must be shipped to our San Francisco lab in an insulated box with frozen cold packs.
USA-based customers
Intact capsids in common virus storage buffers
Our auto-generated UPS shipping labels can only be used for cold pack shipments within the USA. These labels can not be used for dry ice shipments. If you are shipping samples on dry ice you must arrange your own shipping service and ensure the correct hazardous materials labels and handling are observed.
- Place your samples into a 50 mL conical tube.
- Print out your order form. Place the tube in a small bag with a printed copy of your order confirmation sheet (folded so that the QR code is visible).
- Place into an insulated box with frozen cold packs.
- Ship via any Express or First-Class shipping carrier such as FedEx, UPS, DHL, or USPS to:
Plasmidsaurus
2 Tower Place, Suite 950
South San Francisco, CA 94080
Shipping tips
- Dry ice shipping is not required. At this time we're unable to provide shipping for dry ice, and any dry ice shipments must be through your own, or your institution’s, shipping account.
- Do not use one of our dropboxes or one of our UPS shipping labels for dry ice shipping. If you do so, we will not be able to receive your samples.
- Please do not include wet ice in the container since it melts and leaks on to other packages. This can cause the package to be held by the shipping service.
- If you are re-using cold chain boxes with cold packs, be sure to remove all dry-ice placards and hazardous materials warning symbols.
AAV stabilized in DNA/RNA Shield
Dropbox Submission: AAV samples stabilized in Zymo DNA/RNA Shield can be sent through our Dropbox network just like plasmid or PCR samples at ambient temperatures. Our dropbox couriers and regional labs will handle the reshipping to our South San Francisco lab.
Please submit the samples only in a protected 50 mL falcon tube in a ziplock bag. We cannot accommodate boxes or cold pack shipments through dropbox submissions. Do not ship samples already stabilized in DNA/RNA Shield on dry ice.
International AAV shipments must be directly shipped to our South San Francisco lab in the US for processing.
Our auto-generated UPS label does not support dry ice shipping.
Only cold-pack shipping is supported with our auto-generated UPS label.
If the samples are stabilized in Zymo DNA/RNA shield, they can usually be submitted through our dropbox network (please contact support@plasmidsaurus.com to confirm details for your country since specific regulations can vary greatly between countries.)
Dry ice is considered a hazardous material. If samples are shipped on dry ice, customers must generate their own shipping labels through their shipping service of choice and attach the appropriate associated hazardous material labels.
If you are re-using cold chain packaging for cold-pack shipping, please ensure any hazardous materials labels such as checked-diamond dry ice labels are removed. UPS and other shipping services will hold or reject packages if they retain hazardous material labels that are not accounted for during shipping label generation. Plasmidsaurus has no ability to expedite their release or return to customers in these cases since this an issue internal to shipping services.
International packages are often delayed by 3-7 days when shipped to our South San Franisco lab. We strongly recommend submitting AAV stabilized in Zymo DNA/RNA Shield rather than cold chain submissions. In our experience, shipping services that offer dry ice “top up” services often do not sufficiently refill packages to account for extended customs delays.
International Customers shipping intact AAV capsids in common viral storage buffers.
- Due to 3-7 day customs delays, we strongly recommend submitting samples stabilized in DNA/RNA shield. In most cases these samples can be submitted through your local dropbox and the international shipping and customs forms will be handled by our regional labs.
- Cold chain shipments are not accepted through our dropbox system and must be shipped directly to our South San Francisco lab.
AAV submission shipping address:
2 Tower Place
Suite 950
South San Francisco, CA, USA 94080
The auto-generated UPS label can only be used for cold-pack (wet ice) shipments. Dry ice shipments must paid for by the customer and the appropriate hazardous warning labels for dry ice be applied in accordance with the shipping service being used.
Plasmidsaurus is unable to provide labeling or documentation assistance for dry ice shipping. If you are sending a dry ice shipment, please make sure to follow up with your shipping company of choice to ensure you are following their labeling and packaging requirements.
Incorrectly labeled dry ice shipments can result in packages being refused, delayed, or returned. In some cases FAA and similar agency investigations may result due to suspected intentional mislabeling of hazardous materials. Since this is a shipping service matter, Plasmidsaurus has no ability or power to expedite or resolve these problems for our customers.
Please print and fill out the following documents:
- Commercial Shipping Invoice - AAV
For best results, make sure to include the shipper's identity, shipper's address, and the quantity of items within the shipment.
- Detailed Description - AAV
- Toxic Substances Control Act (TSCA) Certificate - AAV
International Customers shipping AAV stabilized in Zymo DNA/RNA shield.
Dropbox Submission of AAV in Zymo DNA/RNA Shield: please contact our support team (support@plasmidsaurus.com) before sending your samples. In most cases our local dropbox network and regional labs can accept your samples but this varies by country. Our regional labs will handle (re)shipping to our South San Francisco lab for sequencing.
Please submit the samples only in a protected 50 mL falcon tube in a ziplock bag. We cannot accommodate boxes or cold-chain shipments through dropbox submissions.
Shipping AAV in DNA/RNA Shield directly to our South San Francisco lab:
Please contact our support team (support@plasmidsaurus.com) to ensure the correct commercial invoice, USDA, and customs forms are included for import in to the USA. In almost all cases, we can generate these forms for you immediately.
Please submit the samples only in a protected 50 mL falcon tube in a larger carboard box to further protect samples.
Although stable at ambient temperatures, cold packs can be added to the shipment provided the samples are in a Styrofoam box inside a cardboard box. UPS will not accept “naked” Styrofoam boxes. Please place insulated boxes inside of a carboard shipping box.
Do not ship samples already stabilized in DNA/RNA Shield on dry ice.
AAV submission should be directed to:
2 Tower Place
Suite 950
South San Francisco, CA, USA 94080
Further guidance for international shipping
- The information below constitutes only a recommendation for shipping samples classified as "non-regulated materials" to our facility. Due to continuing changes in state and federal regulations, clients should always check with their safety office and/or shipping department to ensure regulatory compliance.
- Make sure all forms are fully completed. Incomplete and/or missing forms will likely cause delays and other issues.
- Keep an eye out for any communication or information requests from your shipping company of choice in case of issues. It is your responsibility to provide any information they request, and we advise following their guidance as their systems will have the most up-to-date customs information.
Why do I need these forms? Read more here.
- Samples must be packaged and shipped in accordance with U.S. Department of Transportation (DOT) and/or International Air Transport Association (IATA)/ International Civil Aviation Organization (ICAO) and carrier regulations.
- Your international courier will require you to complete a commercial invoice to be included with the shipment.
- The commercial invoice should describe the contents as “non-hazardous, non-contagious research sample.” Please list your institution as the "Manufacturer" of the samples if Manufacturers Identification (MID) code is required.
- State that the “contents are Not Restricted under IATA DGR regulations.”
- We recommend that you include a Toxic Substance Control Act (TSCA) Certification. If you do not include this, the package may be delayed at customs until the courier receives the proper certification.
- Declare the value of the goods to be $1.
- We only accept AAV samples that are intended for Research Use Only! We DO NOT accept any samples for clinical diagnostics or trials. Any samples that do not conform to our submission guidelines will be discarded and/or submitted to the United States Postal Inspection Service
- If shipping to Cologne, certain sample types will require some additional information. Please reach out to support if you have any questions.
Category B infectious substances (UN 3373) are classified as being potentially harmful to humans or animals and must be packed in accordance with ADR (International carriage of dangerous goods by road) packing instruction PI 650 for road transport and IATA packing instruction PI 650 for air transport. - Unlike other dangerous goods, UN3373 shipments do not require a Shipper's Declaration. However, the Air Waybill (AWB) must include: “UN3373, Biological Substance, Category B” Net quantity
- UN3373 (Biological Substance, Category B) requires triple packaging per Packing Instruction 650 (PI650) to ensure safe transport. This consists of a leakproof primary receptacle, a leakproof secondary packaging with absorbent material, and a rigid outer package, with proper labeling (UN3373 diamond).
- Shipping UN 3373 Biological Substances (Category B) into Germany requires strict adherence to international (IATA for air, ADR for road) and national regulations to ensure safety and compliance with customs. Please make sure you are following these regulations.
- The commercial invoice should describe the contents as “non-hazardous, non-contagious research sample.” Please list your institution as the "Manufacturer" of the samples if Manufacturers Identification (MID) code is required.
- Please make sure to use the correct HTS code to ensure fastest processing. The HTS code we use for our shipments is found on the template commercial invoice document above.
- We recommend including a detailed customs description (the template for which can be found above), or a Material Data Safety Sheet (if you have one already available for your samples). This is not required, but can be requested by customs if a delay occurs.