Submitting Cells Lysed in Zymo DNA/RNA Shield™
This service is available for animal cells or plant protoplasts lysed and preserved in Zymo DNA/RNA Shield™. Zymo DNA/RNA Shield™ promptly lyses cells and stabilizes RNA for up to a month at ambient temperatures. We can't currently accept tissue samples or prokaryotic cells.
Sample quality standards
RNA-seq data quality and accuracy rely on having intact RNA. To minimize RNA degradation, it is important that your sample preparation workflow ensures that your cells are alive, intact, and free of debris.
| Parameter | Recommended Standard |
| Cell viability | ≥ 90% (minimum acceptable: <70% will affect sequence results) |
| Cell aggregates | Minimal to none |
| Debris | Minimal |
To ensure high quality samples, we recommend the following to help assess sample quality prior to submission:
- Use viability dyes to monitor sample health
- Avoid harsh dissociation or extended processing times.
- Add QC checks throughout your sample preparation workflow where possible.
Avoid samples that are:
- Fixed (i.e., formalin, PFA, methanol)
- Heat-damaged
- Highly degraded
Volumes and concentrations
Our automated extraction and library preparation protocols require a minimum of 50 μl of lysate volume containing at least 10 ng/μl of extractable bulk RNA, for a minimum of 500ng of RNA total (1 μg is preferable for best results).
The 50 μl volume requirement is a hard constraint; do not send lower volumes.
500ng is the minimum amount of extractable RNA required to ensure you have a high probability of obtaining the full nominal data quality.
If we are not able to extract a minimum of 10ng/μl concentration from your submitted sample, e.g. due to low transcription levels in your cells or scarcity of available cells, we will process lower concentrations, but read counts obtained may be (substantially) lower.
If you have additional cells available, you may submit more than 50 μl of volume, but you should scale up cell input amounts proportionately. We can only use 50 μl per sequencing run, but if you submit additional volume this can facilitate a rerun in the event of a process issue.
- Do not exceed a cell equivalency of >100 ng/μl RNA concentration or any cell concentration that results in high sample viscosity (see below).
Important sample viscosity considerations
Samples with high cell counts can become viscous after lysis, leading to pipetting failure during automated processing and possible sample loss or underperformance. When submitting lysed cell samples it is very important to avoid high sample viscosity.
If you invert your tube and the sample doesn't move like water, or if it "strings" when you pull a pipette tip away, it is too viscous for automation.
Viscous samples may sometimes be rescued by vigorous, high speed vortexing for two minutes.
If vortexing is not enough to lower viscosity, we suggest you increase the volume of DNA/RNA Shield and try again, noting that RNA concentration will decrease with dilution.
- If you're unable to achieve the necessary RNA concentration without producing viscous samples, consider submitting purified RNA instead of lysed cells.
Typical Cell Input Quantity
The number of cells needed to meet the target RNA input mass (>= 500 ng) can vary widely as a function of the relative transcriptional activity of the cell type you’re using. The following guidance serves as a starting point for estimating the cell number needed. If you think your cells may have substantially lower RNA levels (e.g. resting T-cells) consider performing a test extraction and cell count calibration or submitting a small test order bracketing a range of cell amounts, e.g. 100-400K cells, paying careful attention to the viscosity of samples.
| Cell Type* | Typical # of Cells | Preservative |
|---|---|---|
| Typical cultured cells (e.g. HeLa) | 1-2 x 10^5 | 50 µl Zymo DNA/RNA Shield™ |
| Cells with lower RNA content (e.g. resting lymphocytes, resting T-cells) | 2.5-10 x 10^5 | |
| Cells with high RNA content (e.g. hepatocytes, large polyploid epithelial cells) | 2.5-7.5 x 10^4 |
Safety considerations
If your samples contain pathogens or other potentially toxic agents, we are limited on what we can accept in our labs as shipping and handling of potentially infectious agents is regulated by the CDC. For the following cell types, we can accept only purified RNA, not preserved cells:
- De novo cell line materials sourced from bats or non-human primates
- Whole cell lines from a facility where work with exotic viruses affecting livestock and avian species is conducted
- Animal derived blood, tissue or body parts that come from Bats, non-human primates, or rodents of African origin
- Material from human blood, tissues, and body parts infected with polioviruses, enteroviruses, or serotype H7N9 avian influenza
Please note, Plasmidsaurus is NOT permitted to handle/import any of the following materials in any form (cells or purified RNA):
- Isolated or purified viruses on the Select Agents and Toxin List
- Unpurified and isolated poliovirus, enterovirus, or H7N9 strain avian influenza
Cross-order comparison/large order considerations
If you are interested in comparing data of RNA-Seq samples from two or more different orders OR you are interested in placing an order with >96 samples, we recommend including samples in your order that can be used as anchor samples for batch correction. This batch correction will be made available on your Results page.
Anchor samples are replicates that span multiple batches/orders. Each order can be considered new batch, and our maximum batch size is 96 samples. Therefore, any order that is >96 samples will contain multiple batches.
Requirements for anchor-based batch correction:
- Every batch must include ≥3 anchor samples that each have >5 million unique reads, and at least one other sample to be corrected.
- More anchor samples increases the accuracy of the correction.
- For effective batch correction, your anchor replicates should share the same biology as the groups in your DGE comparison (e.g. your control group). RUV estimates unwanted variation from the anchors — if their expression profile is too distant from the samples being compared, the correction factors it learns may not generalize well to your genes of interest.
- All orders must use the same species reference
Learn more about anchor-based batch corrections in our technical documentation.
Harvest your cells.
For suspension cells:
- Proceed directly to cell counting
For adherent cells:
- Gently wash cells with 1x PBS (phosphate-buffered saline) to remove residual media.
- Detach cells, avoiding prolonged exposure to trypsin.
Optional direct lysis of adherent cells:
Direct lysis of adherent cells in the culture wells can be a superior alternative as it can allow you to exclude any possible influences from trypsinization and avoid laborious and loss-prone pelleting steps. However, as it will generally require more Zymo DNA/RNA Shield™ volume than 50 μl, this approach may not be suitable for low density cultures, or cells with low bulk RNA levels.
- Gently aspirate cell culture medium, wash with cold buffer, and aspirate again to remove the bulk of the liquid volume. Invert the plate and gently tap the plate onto paper towels or wipes on the bench to remove remaining adherent liquid.
- Add the minimum amount of Zymo DNA/RNA Shield™ necessary to completely wet the culture wells, and swirl for thirty seconds or so to mix. A very low speed orbital shaker can be used if available.
- Tilt the plate and collect the lysate carefully. If you note substantial remaining adherent material, you may opt to scrape using a scraper or a pipette tip.
- Proceed to packaging and shipping.
Count and pellet your cells.
- Count cells using a hemocytometer or automated cell counter.
- Centrifuge cells at 300 x g for 5 minutes.
- Carefully aspirate the supernatant.
Resuspend and lyse cells in Zymo DNA/RNA Shield™
Resuspend the cell pellet for a final volume of 50 µL using Zymo 1X DNA/RNA Shield™ (when ordering Zymo DNA/RNA Shield™, you can enter PLASMIDSAURUS at checkout for 10% off).
Do not exceed about 5µl of cell pellet volume (10% of final volume) to avoid creating viscous lysates.
Mix the cells and the Zymo 1X DNA/RNA Shield™ product immediately and thoroughly.
- If the lysed cell solution appears highly viscous, vortex it vigorously for up to 2 minutes to shear chromosomal DNA. See above for important considerations on sample viscosity.
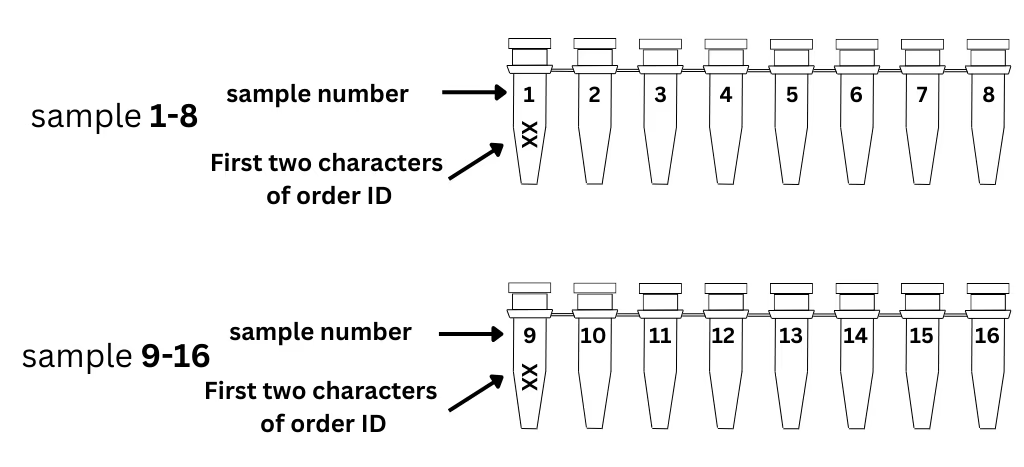
Label your tubes or plates.
- Set up your order online and receive your unique 6-character order ID.
- Note: It can be helpful to include sample metadata (species, tissue/cell type, estimated cell number) in your order notes.
- Write the first 2 characters of your order ID on the 1st tube of each strip
- Set up your order online and receive your unique 6-character order ID. On the order page, please check the “Is this a plate submission?” checkbox, then select if your plate is numbered sequentially by column or by row.
- Note: It can be helpful to include sample metadata (species, tissue/cell type, estimated cell number) in your order notes.
- Label the front edge of the plate with the full 6-character order ID.
- If you are using column numbering, label the first column (i.e., samples 1 to 8). If you are using row numbering, label the first row (i.e., samples 1 to 12).
Load and package your sample.
Add the correct volume of your sample(s) according to the instructions above. Make sure to seal and package your samples well to make sure they arrive safely for sequencing!
- Ensure caps fit snugly and close them firmly. Do not use brands of strip tubes where the caps can easily pop open during shipping.
- Place the labeled tubes into a small protective vessel (such as a Falcon tube, a small box, or bubble wrap) to protect them during shipping. Why?
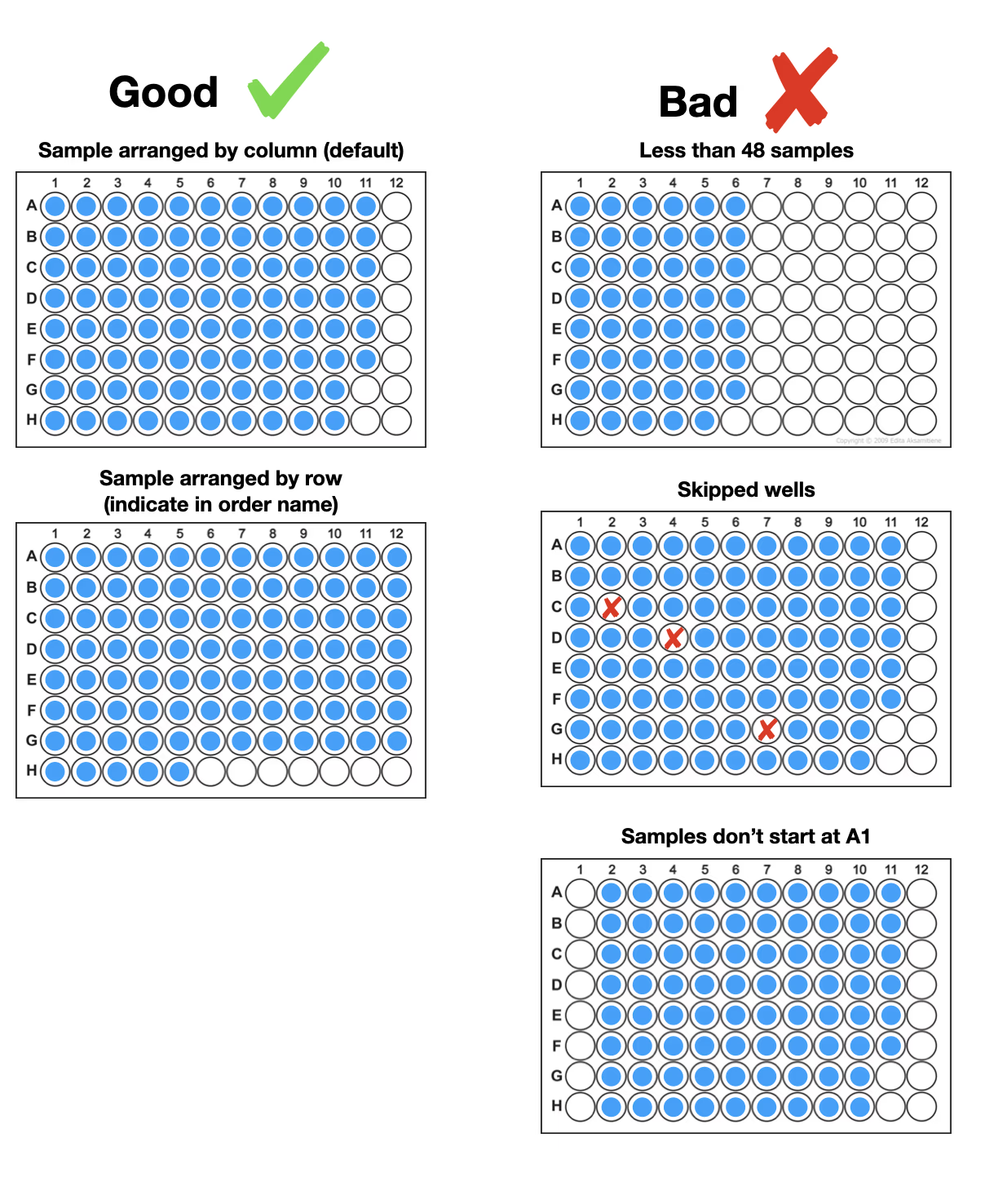
Start with well A1 and load your samples contiguously. Our preference is for samples to be loaded by column but we can accept samples loaded by row if indicated in the order. Do not leave any empty wells between samples.
- Ensure your plates are sealed extremely thoroughly to survive shipping without leaking — sadly we receive a lot of leaky plates. We strongly recommend only using strip caps. For RNA-Seq samples in Zymo DNA/RNA Shield, do not use adhesive seals, as the adhesive will dissolve and the wells will leak.
- Wrap each plate individually in bubble wrap or other packaging material to prevent cracking and seal punctures. DO NOT stack multiple plates directly on top of each other, as the wells of one plate will likely puncture the seal of the next plate.
Dropoff or ship.
Print out your order form. Fold so that QR code is readily visible and place in a small bag with your samples.
For fastest results and easiest shipping, place your samples in a Plasmidsaurus Dropbox!
If you ship your samples to us directly:
- Place packaged samples in a sturdy cardboard or padded envelope or a cardboard box. If reusing old packaging, ensure that any hazard warnings are completely obliterated. Multiple orders can be shipped in the same package, as long as each order is placed into a separate bag with its corresponding order confirmation sheet.
- Do not add ice packs or dry ice. Samples should be shipped at room temperature.
- Ship via any Express or First-Class shipping carrier such as FedEx, UPS, DHL, or USPS. The shipping address will be provided on your order confirmation sheet.
If you aren't using a Plasmidsaurus dropbox, print and fill out the following documents:
- Commercial Shipping Invoice - cell culture
For best results, make sure to include the shipper's identity, shipper's address, and the quantity of items within the shipment.
- Detailed Description - cell culture
- Toxic Substances Control Act (TSCA) Certificate - cell culture (only if shipping from outside of US to US)
Further guidance for international shipping
- The information below constitutes only a recommendation for shipping samples classified as "non-regulated materials" to our facility. Due to continuing changes in state and federal regulations, clients should always check with their safety office and/or shipping department to ensure regulatory compliance.
- Make sure all forms are fully completed. Incomplete and/or missing forms will likely cause delays and other issues.
- Keep an eye out for any communication or information requests from your shipping company of choice in case of issues. It is your responsibility to provide any information they request, and we advise following their guidance as their systems will have the most up-to-date customs information.
Why do I need these forms? Read more here.
- Samples must be packaged and shipped in accordance with U.S. Department of Transportation (DOT) and/or International Air Transport Association (IATA)/ International Civil Aviation Organization (ICAO) and carrier regulations.
- Your international courier will require you to complete a commercial invoice to be included with the shipment.
- The commercial invoice should describe the contents as “non-hazardous, non-contagious research sample.” Please list your institution as the "Manufacturer" of the samples if Manufacturers Identification (MID) code is required.
- State that the “contents are Not Restricted under IATA DGR regulations.”
- We recommend that you include a Toxic Substance Control Act (TSCA) Certification. If you do not include this, the package may be delayed at customs until the courier receives the proper certification.
- Declare the value of the goods to be $1.
- We only accept pure DNA samples that are intended for Research Use Only! We DO NOT accept any samples for clinical diagnostics or trials. Any samples that do not conform to our submission guidelines will be discarded and/or submitted to the United States Postal Inspection Service
- If shipping to Cologne, certain sample types will require some additional information. Please reach out to support if you have any questions.
Category B infectious substances (UN 3373) are classified as being potentially harmful to humans or animals and must be packed in accordance with ADR (International carriage of dangerous goods by road) packing instruction PI 650 for road transport and IATA packing instruction PI 650 for air transport. - Unlike other dangerous goods, UN3373 shipments do not require a Shipper's Declaration. However, the Air Waybill (AWB) must include: “UN3373, Biological Substance, Category B” Net quantity
- UN3373 (Biological Substance, Category B) requires triple packaging per Packing Instruction 650 (PI650) to ensure safe transport. This consists of a leakproof primary receptacle, a leakproof secondary packaging with absorbent material, and a rigid outer package, with proper labeling (UN3373 diamond).
- Shipping UN 3373 Biological Substances (Category B) into Germany requires strict adherence to international (IATA for air, ADR for road) and national regulations to ensure safety and compliance with customs. Please make sure you are following these regulations.
- The commercial invoice should describe the contents as “non-hazardous, non-contagious research sample.” Please list your institution as the "Manufacturer" of the samples if Manufacturers Identification (MID) code is required.
- Please make sure to use the correct HTS code to ensure fastest processing. The HTS code we use for our shipments is found on the template commercial invoice document above.
- We recommend including a detailed customs description (the template for which can be found above), or a Material Data Safety Sheet (if you have one already available for your samples). This is not required, but can be requested by customs if a delay occurs.